A) Atom economy = 28/142 = 20% b) Atom economy = 142/142 = 100% c) If only one product can be sold then the rest is wasted; if both can be sold then there is no waste product and the atom economy is greater. A) Atom economy = 100% b) The reversible reaction symbol ( ) suggests that the reaction is unlikely to have a very high yield. Atom economy and percentage yield are indicators of how efficient a chemical reaction is. Atom economy is the second principle of green chemistry. The ideal atom economy, known as 100% atom economy, for a chemical reaction is taken as the process where all reactant atoms are found in the desired product. In other words, atom economy is a.
Next up in our twelve principles of green chemistry series, we are talking atom economy! Much like the previous piece focusing on waste prevention, this piece will give an overview of atom economy and how it relates to green chemistry. Keep reading to recap our previous principle and learn about the second principle of green chemistry!
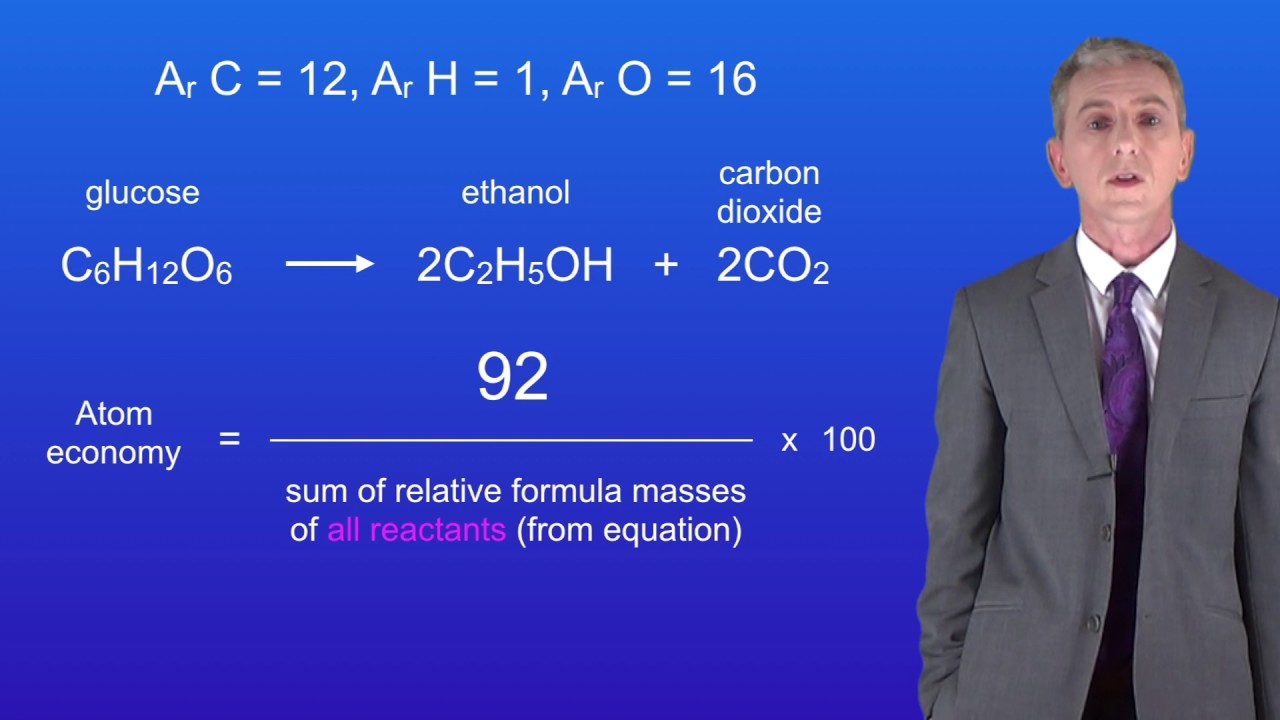
- Atom economy can be written as:% atom economy = (Molecular weight of desired product ÷ Molecular weight of all reactants) × 100 Atom economy can be poor even when chemical yield is near 100%. Atom economy can also be adjusted if a pendant group is recoverable. However, if this can be avoided it is more desirable, as.
- Describes the number of molecules in a compound and is used to balance a chemical reaction.
Recapping the Twelve Principles of Green Chemistry & Waste Prevention
In case you missed our first twelve principles of green chemistry blog, we will briefly inform you about what they are and what the first principle is. The twelve principles of green chemistry are beliefs intended to reduce the chemical impact on human health and environmental contamination by creating more sustainable practices and programs.
The previous blog covered waste prevention. To sum it up, waste prevention as a principle of green chemistry is exactly what you think it is. Preventing waste instead of handling it after its creation. With that mindset, recycling and reusing aren’t the main priorities. We also talked about common misconceptions of waste prevention. Click here to check out the article if you missed it.
What is Atom Economy?
Normally those two words are not words seen together. Atoms are more scientific whereas the economy deals with money and politics. However, when you combine them you create the second principle of green chemistry.
Atom economy asks the question, what atoms of the reactants are incorporated into the final desired product(s) and what atoms are wasted? Another way of looking at this is how much of what you put in your pot ends up in your meal.
Atom Economy in Expanded Use
Atom economy is pollution control at the molecular level. Just as we mentioned in our previous principles of green chemistry blog, waste works in many different ways whether it is physical garbage waste, waste in the air and beyond!
A great example of a chemist practicing atom economy is choosing to synthesize a needed product by putting together basic materials needed instead of breaking down a much larger material for components and discarding a majority as waste.
Anyone can determine the atom economy percentage by using a simple formula. The formula is the mass of the desired product divided by the total mass of the reagents multiplied by 100. This number will be a whole number so just add the percent sign afterward to get the full percentage.
Avomeen’s Commitment to Green Chemistry
Protecting the environment and following the twelve principles of green chemistry is something we take seriously. Avomeen has a proven track record of helping to develop and reformulate greener, more environmentally friendly products for our clientele. In addition to helping clean the products up, we have improved the efficacy to meet or beat important competitor’s levels. Our consultative team of formulators would love to partner with you on a future project. If you have something in mind, reach out, and we’d be happy to discuss a partnership with you.
definition - atom economy
definition of Wikipedia
Advertizing ▼
Wikipedia
Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted. Recent developments like high raw material (such as petrochemicals) costs and increased sensitivity to environmental concerns have made atom economical approaches more popular. Atom economy is an important concept of green chemistry philosophy,[1][2][3] and one of the most widely used ways to measure the 'greenness' of a process or synthesis.
Atom economy can be written as:
Atom Economy Ppt
% atom economy =
Note that atom economy can be poor even when chemical yield is near 100%, see for instance the Cannizzaro reaction or the Wittig reaction. If the desired product has an enantiomer the reaction needs to be sufficiently stereoselective even when atom economy is 100%. A Diels-Alder reaction is an example of a potentially very atom efficient reaction that also can be chemo-, regio-, diastereo- and enantioselective. Catalytic hydrogenation comes the closest to being an ideal reaction that is extensively practiced both industrially and academically.[4] The Gabriel synthesis of amines is an example of extremely low atom economy, as stochiometric quantities of phthalic acid derivatives are formed. In most cases, the atom economy of the Gabriel synthesis is <<50%.
Atom economy can also be adjusted if a pendant group is recoverable, for example Evans auxiliary groups. However, if this can be avoided it is more desirable, as recovery processes will never be 100%. Atom economy can be improved upon by careful selection of starting materials and a catalyst system.
Atom economy is just one way to evaluate a chemical process. Other criteria can include energy consumption, pollutants released and price.
Poor atom economy is common in fine chemicals or pharmaceuticals synthesis, and especially in research, where the aim to readily and reliably produce a wide range of complex compounds leads to the use of versatile and dependable, but poorly atom-economical reactions. For example, synthesis of an alcohol is readily accomplished by reduction of an ester with lithium aluminum hydride, but the reaction necessarily produces a voluminous floc of aluminum salts, which have to be separated from the product alcohol and disposed of. The cost of such hazardous material disposal can be considerable. Catalytic hydrogenolysis of an ester is the analogous reaction with a high atom economy, but it requires catalyst optimization, is a much slower reaction and is not applicable universally.
Creating reactions utilizing atom economy
It is fundamental in chemical reactions of the form A+B→ C+D that two products are necessarily generated though product C may have been the desired one. That being the case, D is considered a byproduct. As it is a significant goal of green chemistry to maximize the efficiency of the reactants and minimize the production of waste, D must either be found to have use, be eliminated or be as insignificant and innocuous as possible. With the new equation of the form A+B→C, the first step in making chemical manufacturing more efficient is the use of reactions that resemble simple addition reactions with the only other additions being catalytic materials.
Atom Economy Means
References
Atom Economy Calculation
- ^Trost B. M. (1995). 'Atom Economy. A Challenge for Organic Synthesis'. Angew. Chem. Int. Ed. Engl.34 (3): 259–281. DOI:10.1002/anie.199502591.
- ^Sheldon R. A. (2000). 'Atom efficiency and catalysis in organic synthesis'. Pure and Applied Chemistry72 (7): 1233–1246. DOI:10.1351/pac200072071233. http://www.iupac.org/publications/pac/2000/7207/7207pdf/7207sheldon_1233.pdf.
- ^Atom Economy: A Green Chemistry Module.
- ^Solaza, B.; Huguet, J.; Karpf, M. and Dreiding, A. S. (1987). 'The Synthesis of (+/-)Isoptychanolide by Application of the a-Alkynone Cycilsation'. Tetrahedron43 (21): 4875–4886. DOI:10.1016/S0040-4020(01)87670-5.
Atom Economy Green Chemistry
Retrieved from 'http://en.wikipedia.org/w/index.php?title=Atom_economy&oldid=493956711'
This entry is from Wikipedia, the leading user-contributed encyclopedia. It may not have been reviewed by professional editors (see full disclaimer)
